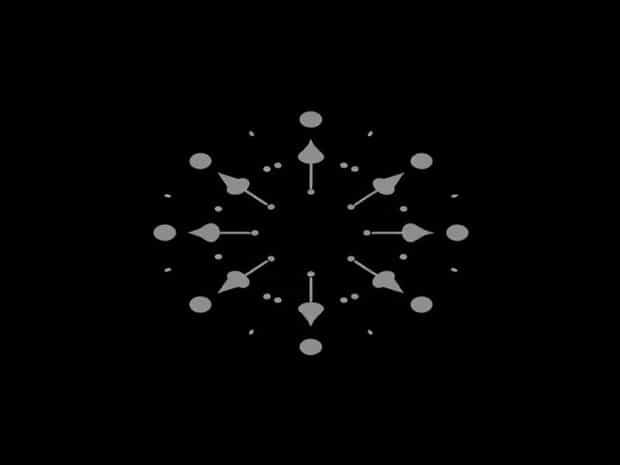
Lewis Dot Structure For Phosphorus
Understanding the Lewis dot structure for phosphorus is a fundamental concept in chemistry, particularly in the study of chemical bonding and molecular geometry. Phosphorus, a group 15 element on the periodic table, has five valence electrons that play a crucial role in forming covalent bonds with other atoms. Drawing the Lewis dot structure helps visualize how phosphorus shares electrons, achieves stability, and participates in various chemical compounds. This knowledge is essential for students, chemists, and anyone interested in predicting molecular behavior, reactivity, and the formation of complex phosphorus-containing molecules.
Basics of Lewis Dot Structures
Lewis dot structures, also known as electron dot structures, are diagrams that represent the valence electrons of an atom or molecule. Valence electrons are the outermost electrons that determine an element’s chemical reactivity. In a Lewis dot structure, dots are placed around the chemical symbol to indicate these electrons. Shared electron pairs between atoms are represented as lines or pairs of dots, showing covalent bonds. Understanding these structures is essential for predicting molecular geometry, bond angles, and reactivity patterns in chemical compounds.
Valence Electrons in Phosphorus
Phosphorus has the electron configuration [Ne] 3s² 3p³, giving it five valence electrons in the third shell. These electrons are available for bonding with other atoms. In the Lewis dot structure, these five electrons are represented as five dots arranged around the symbol ‘P’. The arrangement typically follows Hund’s rule, with one electron in each of the three p orbitals before pairing occurs. This configuration allows phosphorus to form three single covalent bonds, or in some cases, five bonds in compounds like phosphorus pentachloride (PCl₅).
Drawing the Lewis Dot Structure for Phosphorus
To draw the Lewis dot structure for phosphorus, follow a few simple steps. First, write the symbol ‘P’ for phosphorus. Then, determine the number of valence electrons, which is five for phosphorus. Place these electrons around the symbol in pairs or single dots to represent unpaired and shared electrons. The distribution of electrons is important for predicting how phosphorus will bond with other atoms and the resulting molecular geometry. Visualizing electron placement is crucial for understanding chemical stability and reactivity.
Single Atom Lewis Dot Structure
For a single phosphorus atom, the Lewis dot structure consists of the symbol ‘P’ surrounded by five dots. These five dots are arranged to maximize separation and minimize repulsion, often with three dots on three sides and a pair of dots on the fourth side. This arrangement reflects the three unpaired electrons available for bonding and the lone pair that remains on the atom. Recognizing this lone pair is important when predicting molecular shapes using VSEPR (Valence Shell Electron Pair Repulsion) theory.
Phosphorus in Compounds
Phosphorus commonly forms compounds such as phosphine (PH₃), phosphorus trichloride (PCl₃), and phosphorus pentachloride (PCl₅). In PH₃, phosphorus forms three single covalent bonds with hydrogen atoms, using three of its valence electrons. The remaining two electrons form a lone pair, which influences the molecular shape and bond angles. In PCl₅, phosphorus expands its octet and uses all five valence electrons to form five bonds with chlorine atoms. The ability to form more than an octet is characteristic of elements in the third period and beyond, including phosphorus.
Electron Pair Geometry and Molecular Shape
The Lewis dot structure of phosphorus not only shows the bonding electrons but also helps predict the three-dimensional geometry of molecules. The VSEPR theory states that electron pairs around a central atom arrange themselves to minimize repulsion. For example, in PH₃, the three bonded pairs and one lone pair create a trigonal pyramidal shape. In PCl₅, with five bonded pairs and no lone pairs, the molecular geometry is trigonal bipyramidal. Understanding these geometries is crucial for predicting chemical reactivity and physical properties.
Resonance and Multiple Bonding
While phosphorus primarily forms single bonds in most compounds, it can also participate in multiple bonding in certain molecules, such as phosphorus oxychloride (POCl₃) and phosphate ions (PO₄³⁻). In these cases, double bonds may form, and resonance structures can be drawn to depict electron delocalization. The Lewis dot structure is flexible enough to represent these variations, helping chemists understand bond order, electron distribution, and molecular stability. Resonance and multiple bonding play a key role in the chemical behavior of phosphorus-containing compounds.
Common Mistakes in Drawing Lewis Structures for Phosphorus
Many students make errors when drawing Lewis dot structures for phosphorus. A common mistake is underestimating the ability of phosphorus to expand its octet, leading to incomplete representations in compounds like PCl₅. Another mistake is misplacing the lone pair, which can affect predictions of molecular geometry. Ensuring the correct number of valence electrons and proper placement around the atom is essential for accurate Lewis structures. Practicing with multiple phosphorus-containing molecules helps build proficiency and confidence.
Tips for Accurate Lewis Structures
- Always count valence electrons before drawing the structure.
- Place single electrons first, then pair electrons as needed.
- Consider lone pairs and their effect on molecular geometry.
- For elements in the third period or higher, remember that expanded octets are possible.
- Check total electron count to ensure it matches the sum of valence electrons.
Applications of Phosphorus Lewis Structures
Lewis dot structures for phosphorus are widely used in chemistry education, research, and industry. They help in predicting chemical reactivity, understanding acid-base behavior, and designing new compounds in pharmaceuticals and materials science. For instance, knowing the electron configuration and bonding capacity of phosphorus aids in synthesizing organophosphorus compounds, which are used in fertilizers, flame retardants, and biochemical applications. Accurate Lewis structures are essential for modeling chemical reactions and understanding reaction mechanisms.
Phosphorus in Biological Systems
In biology, phosphorus is a key element in DNA, RNA, ATP, and phospholipids. The Lewis dot structure helps illustrate how phosphorus forms bonds with oxygen in phosphate groups, contributing to energy transfer and genetic information storage. Understanding the bonding patterns and lone pairs of electrons in phosphorus is fundamental to biochemistry, molecular biology, and enzymology. This knowledge enables scientists to predict how phosphorus interacts in metabolic pathways and cellular structures.
The Lewis dot structure for phosphorus provides a clear and visual representation of its valence electrons, bonding potential, and lone pairs. By understanding this structure, students and chemists can predict molecular geometry, reactivity, and chemical properties of phosphorus-containing compounds. From simple molecules like PH₃ to complex ions like PO₄³⁻, the Lewis dot structure is an invaluable tool for understanding how phosphorus interacts in chemical and biological systems. Mastery of Lewis structures not only strengthens foundational chemistry skills but also supports advanced studies in molecular design, biochemistry, and materials science, highlighting the versatility and significance of phosphorus in science.