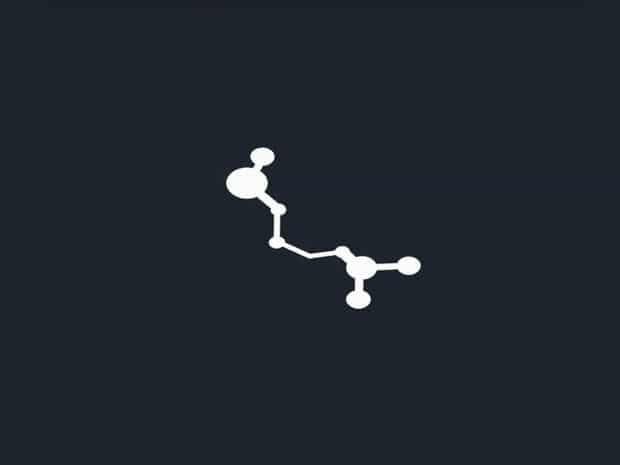
Molecular Formula Of Guanine
Guanine is one of the fundamental building blocks of nucleic acids, making it a crucial component of DNA and RNA. It is classified as a purine base and plays a significant role in the storage and transfer of genetic information. Understanding the molecular formula of guanine is essential for students, researchers, and anyone interested in molecular biology, biochemistry, or genetics. The structure, chemical composition, and properties of guanine contribute to its ability to form hydrogen bonds with cytosine, which is vital for the stability and replication of DNA.
What is Guanine?
Guanine is an organic compound and one of the four nucleobases in DNA, alongside adenine, cytosine, and thymine. In RNA, uracil replaces thymine, but guanine remains a critical base. As a purine nucleobase, guanine consists of a two-ring structure a fused imidazole and pyrimidine ring. This structure allows guanine to engage in specific hydrogen bonding with cytosine, forming three hydrogen bonds that help stabilize the DNA double helix. Guanine is also involved in various biochemical processes, including energy transfer, signaling, and protein synthesis.
Molecular Formula of Guanine
The molecular formula of guanine isC5H5N5O. This formula indicates that a single molecule of guanine contains five carbon atoms, five hydrogen atoms, five nitrogen atoms, and one oxygen atom. The nitrogen-rich structure is characteristic of purine bases and is responsible for its ability to participate in hydrogen bonding, contributing to the fidelity of genetic information during DNA replication.
Structural Features
Guanine’s structure can be described as a bicyclic system, combining a six-membered pyrimidine ring and a five-membered imidazole ring. Key structural features include
- Five nitrogen atoms positioned at strategic locations in the rings, enabling hydrogen bonding.
- A carbonyl group (C=O) at the C6 position, which participates in hydrogen bonding with cytosine.
- An amino group (-NH2) at the C2 position that enhances base pairing and molecular recognition.
These structural elements are critical for the formation of the guanine-cytosine base pair and contribute to the overall stability of nucleic acid structures.
Role of Guanine in DNA and RNA
In DNA, guanine pairs specifically with cytosine through three hydrogen bonds, which makes the G-C pair more stable than the A-T pair, which has only two hydrogen bonds. This stability is particularly important in regions of the genome with high G-C content, as it can affect the melting temperature of DNA and influence gene expression. In RNA, guanine also pairs with cytosine, contributing to the formation of secondary structures like hairpins and loops that are essential for RNA function.
Biochemical Importance
- Nucleotide FormationGuanine combines with a ribose or deoxyribose sugar and a phosphate group to form nucleotides, which are the monomers of nucleic acids.
- Energy MetabolismGuanine derivatives, such as guanosine triphosphate (GTP), are involved in energy transfer, signal transduction, and protein synthesis.
- Cell SignalingCyclic GMP, a derivative of guanine, acts as a second messenger in several biological signaling pathways, regulating processes such as vision and vasodilation.
Physical and Chemical Properties
The chemical composition indicated by the molecular formulaC5H5N5Oalso determines the physical and chemical properties of guanine. It is relatively insoluble in water but soluble in dilute acids and alkalis. Guanine has a high melting point due to its rigid structure and extensive hydrogen bonding. These properties make guanine a stable molecule under physiological conditions, allowing it to maintain its integrity in DNA and RNA molecules over long periods.
Hydrogen Bonding and Base Pairing
Guanine forms three hydrogen bonds with cytosine, which involve the following interactions
- The amino group at the C2 position of guanine donates a hydrogen to the carbonyl oxygen of cytosine.
- The carbonyl oxygen at the C6 position of guanine accepts a hydrogen from the amino group of cytosine.
- The nitrogen at the N1 position of guanine accepts a hydrogen from the N3 position of cytosine.
This triple hydrogen bonding contributes significantly to the thermodynamic stability of DNA and ensures accurate replication of genetic material.
Synthetic and Natural Sources
Guanine is found naturally in all living organisms as part of DNA and RNA. It can also be synthesized chemically for research and medical purposes. Natural sources of guanine include fish, meat, legumes, and certain vegetables. In scientific research, guanine is often studied in its isolated form or as part of nucleotides to understand its role in genetic coding, molecular recognition, and enzymatic reactions.
Applications of Guanine
- Genetic ResearchStudying guanine and its molecular formula helps scientists understand DNA replication, mutation, and repair mechanisms.
- Drug DevelopmentGuanine analogs are used in antiviral and anticancer therapies by interfering with nucleic acid synthesis.
- BiotechnologyGuanine derivatives are used in molecular biology techniques, including PCR and sequencing, for accurate genetic analysis.
The molecular formula of guanine, C5H5N5O, represents more than just a combination of atoms; it reflects a highly functional molecule that is central to life. Guanine’s nitrogen-rich, bicyclic structure allows it to form stable hydrogen bonds with cytosine, ensuring accurate genetic coding and replication. Beyond its role in DNA and RNA, guanine participates in energy metabolism, signaling pathways, and enzymatic reactions. Understanding the molecular formula and properties of guanine is crucial for researchers, students, and healthcare professionals, offering insights into genetics, biochemistry, and biotechnology. Its stability, biochemical versatility, and structural importance make guanine an indispensable molecule in all living organisms.